



|
||||||||
| Genomic Services | ||||||||
|
||||||||
| Protein Services | ||||||||
|
||||||||
Located in rooms
B065 and B017 |
| Mass Spectrometry |
• OVERVIEW |
INSTRUMENT
Perseptive Biosystems(ABI) - Voyager-DE RP - MALDI-TOF mass spectrometer is equipped with Delayed Extraction technology (DE),
which provides improved resolution, sensitivity, and mass accuracy. It is capable of analyzing the intact mass of peptides, proteins*, oligonucleotides, polymers, and small molecules.
DESCRIPTION |
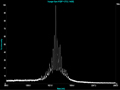
MASS RANGE
The instrument is capable of measuring mass over the range of 100Da
to approximately 200,000Da. However, below 500Da, the matrix
ions may obscure the sample mass, and above 100,000Da, ionization is
difficult. In linear mode, the instrument has an optimal working range
of 500Da to 100,000Da. In reflector mode, the instrument has an optimal working range of 500Da to 4000Da.
SENSITIVITY
High quality spectra can be obtained from 1uL of 100fmol-10pmol of sample loaded onto the target plate. Too much material
can broaden spectral peaks, resulting in lower resolution, and in extreme cases may suppress ionization.
The presence of excess salts at concentrations
greater than 50mM can also cause significant ion suppression.
ACCURACY
Mass accuracies depend on calibration of the instrument against
a standard, and also upon the condition of the sample. Too much
starting material, high salt content, or the presence of glycerol,
detergents and urea can broaden spectral peaks, and compromise mass
accuracy. Typical values when calibrating against an external mass
standard for accuracy are 0.1% (1Da per 1000Da) in linear mode, and 0.01% (1Da per 10,000Da) in reflector mode.
TURNAROUND TIME
We expect to return spectra to the investigator within 72 hours of
receiving the samples.
Home | FAQs | | Prices | Contact Us | Publications | Feedback
Beckman Center | Stanford Medical Center | Stanford University
© 2006 Stanford PAN Facility. All rights reserved
